How do I take part in a clinical study?
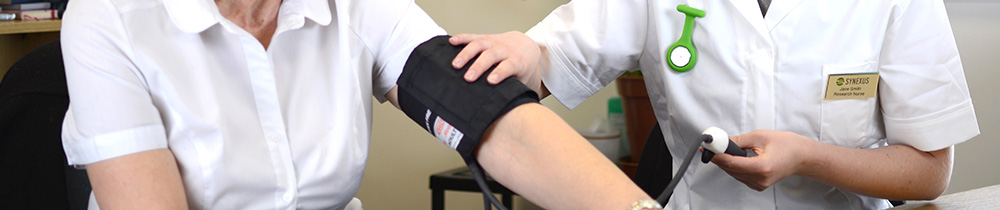
Taking part in a clinical study is a personal choice and we will provide the information you need to make an informed decision. We encourage you to discuss your decision with those close to you as well.
If you decide you’re interested in potentially taking part, we make your journey as simple and informative as possible:
When you register from any page on this website, we will ask you some questions to see if you’re suitable for one of our studies because it is important that we learn more about you so we can find the right study for you.
If you’re eligible a member of our team will explain any benefits and risks in detail and provide you an Informed Consent Form.
You can take this form home to discuss it with your family before making a decision.
If you decide to take part, and sign the informed consent form, you are given a simple treatment plan to follow. Your condition will be monitored throughout the study and you will be offered specialist advice and support throughout.
The plan may require you to visit our clinic regularly, and we may be able to help with your travel arrangements. You are also welcome to bring someone with you to the clinic for support at any time.
Remember you can express an interest without committing to a study and you are free to withdraw at any time without giving a reason.
Find a clinical study that's right for you
Register your interest and a member of our team will contact you.
 Call Synexus Now
Call Synexus Now
 Bulgaria
Bulgaria Czech Republic
Czech Republic Germany
Germany Hungary
Hungary Poland
Poland South Africa
South Africa Ukraine
Ukraine United Kingdom
United Kingdom















